Spirited display: the first study to show that DMT protects brain and immune cells under stress
Since the discovery of endogenous DMT (when the human body produces the chemical of its own accord) by the Hungarian psychiatrist Stephen Szára in 1956, scientists have been entranced by the “spirit molecule” and have been determined to find out how and why it produces its psychedelic effects, which are so repeatedly likened to “near-death experiences” by study participants; as Rick Strassman famously explored, recording the other-worldly trips of 400 subjects between the years of 1990 and 1995; and recreational users alike.
Their explanations ranged from the action of DMT having a purely physiological basis- that producing DMT is the body’s way of trying to regulate neural signalling and brain/peripheral immunological processes (Shen et.al., 2010; Frecska et al., 2013; Szabo et al. 2014) to more spiritual suggestions: that DMT is enabling communication with the divine.
A new study – aptly from a Hungarian team- led by Ede Frecska, and our collaborator Jordi Riba, is the first to bring some clarity to the mystique surrounding this hotly-debated subject.
The scientific set-up: the team took 3 types of human cells – neurons derived from pluripotent stem cells (a cell that can develop into any other cell type), monocyte-derived macrophages and dendritic cells (these latter two are both key players in the body’s immune system, working to engulf and annihilate undesirable particles, such as harmful bacteria or cancer cells). The team then restricted the cells’ supply of oxygen (hypoxia). A few hours of severe hypoxia reliably kills the majority of neurons, and a significant proportion of the immune cells.
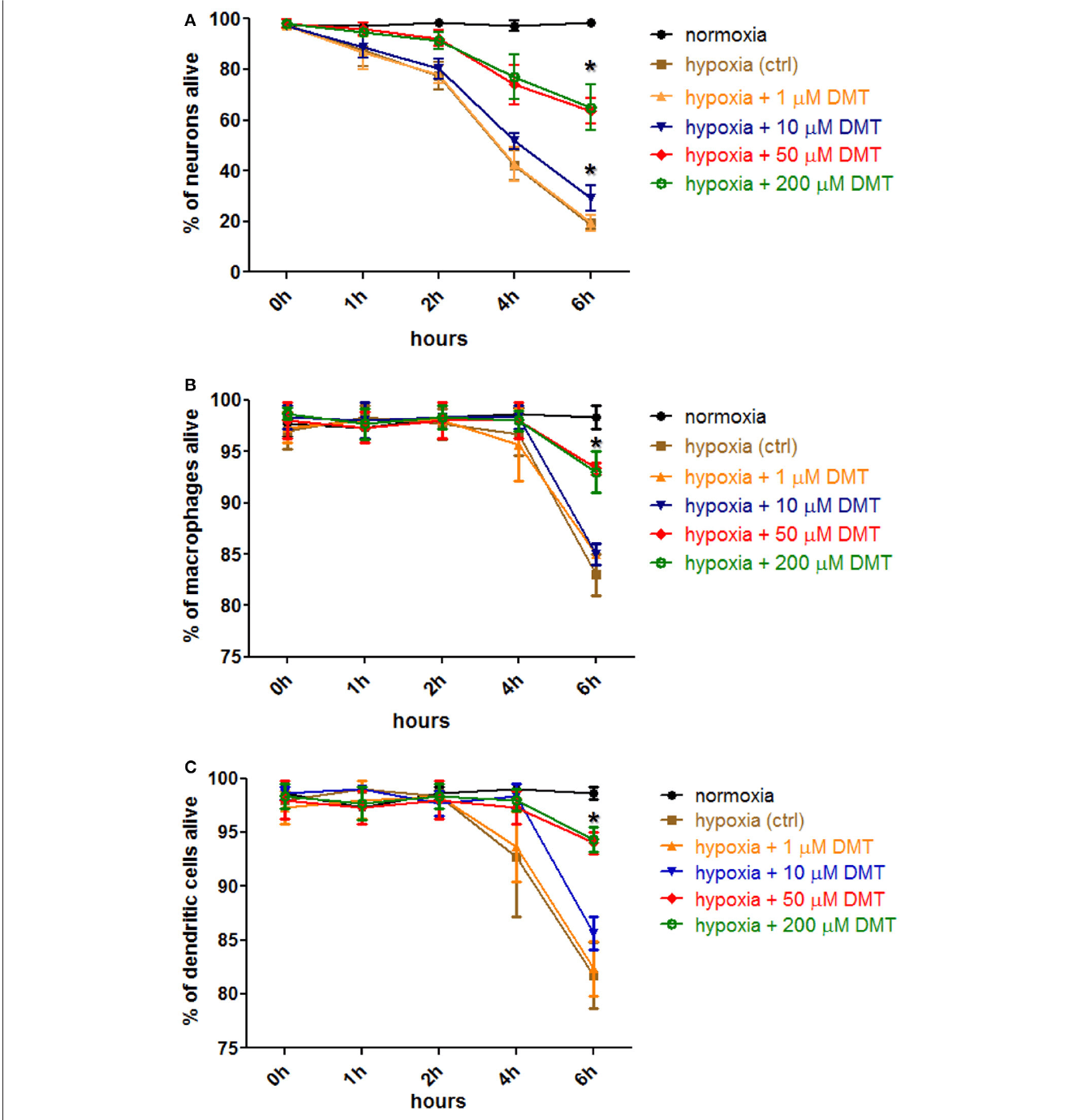
Enter, the spirit-molecule: The scientists then tested whether DMT could protect the cells from this fate. And indeed, they found that DMT-treated cell cultures had a much higher survival rate! Moreover, the amount of DMT needed to protect the cells was relatively low (10-50μM for neurons and 50 μM for the dendrites and macrophages) – see figure below.
FIGURE 2 | The effect of DMT treatment on the survival of human cortical neurons, monocyte-derived macrophages and dendritic cells in hypoxic environment. Hypoxia treatment (0.5% O2) of cell cultures and cellular viability assays were carried out. Prior to hypoxia treatment, human primary cortical neurons (A), moMACs (B), and moDCs (C) were either treated with 1–200μM DMT or left untreated (hypoxia control). Normoxic cultures were used as positive controls (normoxia). Induction of cellular death was assessed by Annexin V-FITC staining. Flow cytometry data of three (neurons) or six (macrophages and dendritic cells) independent experiments are shown as Mean ± SEM. In each case, asterisk indicates significance compared to hypoxia controls (p < 0.05). Modified from Szabo et al., 2016
Hypoxia usually causes dramatic alterations to unprotected cells. One of the most reliable markers of being oxygen-deprived –the hypoxic condition- is the presence of the so-called hypoxia-inducible factor (HIF)-1, which can be clearly visualised (see figure below), but disappears when there is sufficient amount of oxygen. In the presence of DMT, there was significantly less HIF-1 detected in the cells, as well as the other oxygen-sensitive proteins. Therefore, DMT could be seen to actively reduce cellular stress in hypoxic environments.
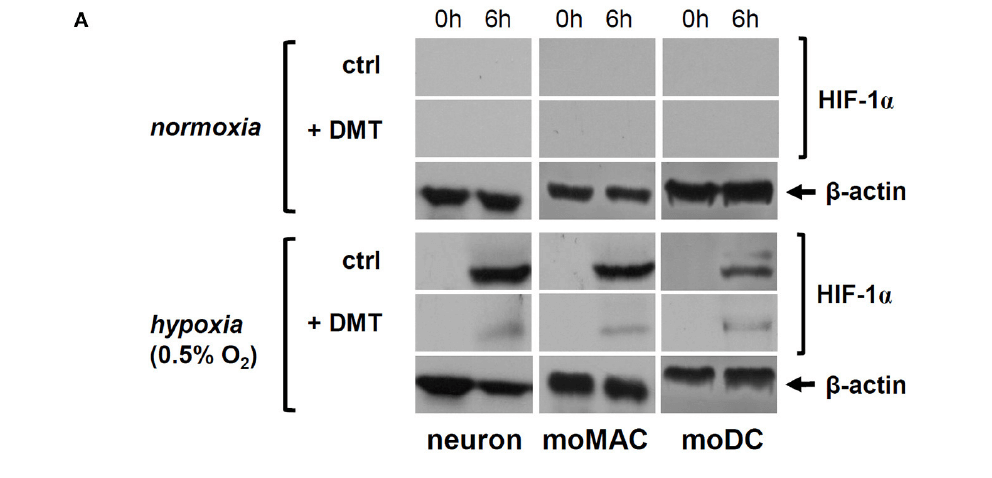
FIGURE 2 | DMT interferes with the expression and function of HIF-1a in human primary cells. (A) Prior to their placement into hypoxia chamber (0.5% O2), cells were treated with 50 μM DMT (+DMT) or left non-treated (controls). Protein level expression of HIF-1a was evaluated by Western blotting after 6 h of hypoxia treatment (6 h) in comparison with the baseline expression (0 h). A control experiment was performed at normal oxygen level (normoxia). Modified from Szabo et al., 2016
So, the DMT-treated cells underwent –on a, literally, microcosmic level– a “near-death” experience, as opposed to a deadly one. The next research question is: how does DMT protect these cells?
Szabo et al. decided to investigate how important the action of the sigma-1 receptors (Sig-1R) is in bringing about the protective effects of DMT. Located inside the immune and brain cells (and cells from other organs, such as liver, kidney or gut), these receptors have important functions in the synthesis of energy molecules, as well as facilitating stress signalling. On a molecular level, “stress,” does not correspond to the difficulties and hurdles of everyday life, but to all the nasty things that threaten a cell’s well-being, such as changes in oxygen level (used in this study), temperature, toxins and mechanical damage. DMT has been previously shown to bind to Sigma-1 receptors (Fontanilla et al., 2009), and more DMT is detected in the central nervous system under stress (Barker et al., 1981; Fontanilla et al., 2009).
Suspecting that the effects of DMT hinge on the action of these receptors, the team selectively silenced (i.e. switched off) the genes responsible for SIG-1R development. The results were drastic and immediately apparent: switching of the sigma-1 receptors led to complete annihilation of the DMT-related effects on cell survival and HIF-1 markers! Therefore, Szabo et al. conclude that sig-1R are absolutely instrumental in enabling the protective, anti-stress effects of DMT.
By finding out that these effects are mediated through the Sig-1 receptors, Szabo et al. have brought us one step closer to finding out mechanisms behind DMT’s neuroprotective effects. We can look more confidently to a future where the workings of DMT are better understood. Currently plausible hypotheses include the idea that DMT is fine tuning the regulation of oxygen metabolism, or relating to other molecules involved in cell survival.
The authors conclude: “Our results suggest a novel and important role of DMT in human cellular physiology and point out to the relevance of DMT-mediated Sig-1R modulation in future therapies concerning hypoxia/ischemia-related pathologies.” If these results are applicable to other stressors and replicated in humans, the implications are huge. DMT could be administered to save neurones that would otherwise die from exposure to harmful events, such as to protect patients’ brains against damage caused by oxygen starvation – e.g. during heart attack or stroke. And the damage wrought by tragic neurodegenerative diseases, like Parkinson’s and Alzheimer’s –for which there are currently no cures– could potentially be redressed. On a biological level, as well as a cosmic one (as some people believe), DMT could be a life-saving agent, administered in order to give cells, and the bodies they belong to, a second chance at life.
Words: Anna Ermakova and Rosalind Stone
Podcast
- All
Links
- All
Support
- All
BIPRP
- All
Science Talk
- All
Amanda's Talks
- All
- Video Talk
- Featured
- 2016 Onwards
- 2011-2015
- 2010 and Earlier
- Science Talk
- Policy Talk
One-pager
- All
Music
- All
Amanda Feilding
- All
Events
- All
Highlights
- All
Psilocybin for Depression
- All
Current
- All
Category
- All
- Science
- Policy
- Culture
Substance/Method
- All
- Opiates
- Novel Psychoactive Substances
- Meditation
- Trepanation
- LSD
- Psilocybin
- Cannabis/cannabinoids
- Ayahuasca/DMT
- Coca/Cocaine
- MDMA
Collaboration
- All
- Beckley/Brazil Research Programme
- Beckley/Maastricht Research Programme
- Exeter University
- ICEERS
- Beckley/Sant Pau Research Programme
- University College London
- New York University
- Cardiff University
- Madrid Computense University
- Ethnobotanicals Research Programme
- Freiburg University
- Medical Office for Psychiatry and Psychotherapy, Solothurn
- Beckley/Sechenov Institute Research programme
- Hannover Medical School
- Beckley/Imperial Research Programme
- King's College London
- Johns Hopkins University
Clinical Application
- All
- Depression
- Addictions
- Anxiety
- Psychosis
- PTSD
- Cancer
- Cluster Headaches
Policy Focus
- All
- Policy Reports
- Advisory Work
- Seminar Series
- Advocacy/Campaigns
Type of publication
- All
- Original research
- Report
- Review
- Opinion/Correspondence
- Book
- Book chapter
- Conference abstract
- Petition/campaign
Search type